Biocalcification and Carbonate Geochemistry
Carbonate skeleton and tests of marine calcifiers are among the most important environmental archives, because their elemental and isotopic compositions reflect the chemical and physical environments from which they formed. This constitutes the basis of all geochemical proxies in biogenic carbonates. We study the mechanism of biocalcification, with the goal of more accurately interpreting these geochemical proxies and of predicting the response of biocalcification to global change and ocean acidification.
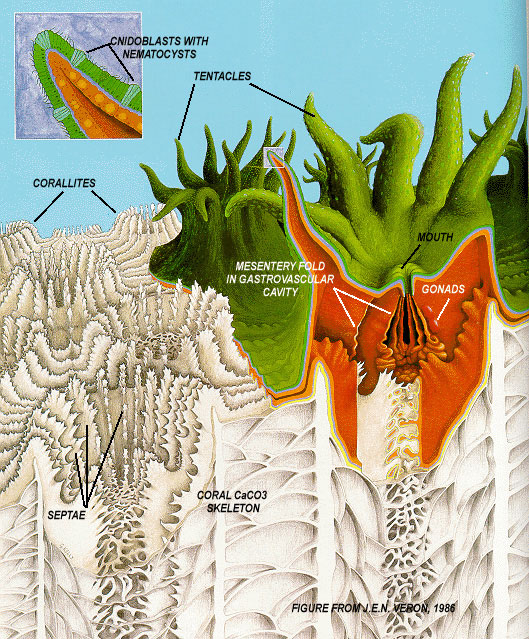
Modeling pH Regulation During Coral Calcification: Implications for Predicting Coral Calcification Responses to Ocean Acidification and for Interpreting Geochemical Proxies
Coral, like many other carbonate-secreting organisms, exerts strong control over its calcification process, resulting in an internal calcifying environment that departs from the ambient seawater. Most notably, pH of the coral calcifying fluid has been shown to be significantly elevated relative to seawater. Such pH regulation not only determines coral calcification responses to environmental changes (e.g. ocean acidification and rising temperature), but also affects the elemental and isotopic compositions of coral skeleton and complicates the interpretation of various geochemical proxies. Based on existing models of coral calcification and geochemical constraints on its pH regulation, I simulate the chemistry, especially pH, of the coral calcifying fluid, evaluate the calcification responses to changing seawater chemistry among different coral species, and explore the implication of these model results for interpreting geochemical proxies. (Guo 2014).

Simulating Coral Calcification in the Lab: Impact of Ocean Acidification and Rising Temperature
Coral reefs form some of the most diverse ecosystems on Earth and have tremendous ecological and economic values. As the key process to the health of these ecosystems, coral calcification is affected by the ocean acidification and rising temperature induced by anthropogenic CO2 emission. Most of our current understanding of the impact of these environmental changes on coral reef calcification derives either from laboratory manipulation experiments in which corals are removed from natural habitat and reared under controlled conditions or from in situ measurements of calcification response of selected reef ecosystem. We are conducting chemical precipitation experiments to simulate coral calcification process in the laboratory, determine ‘calcification’ rates under different temperature and pCO2/pH conditions, and quantitatively evaluate the effects of several key variables of coral calcification on its calcification rates. Supported by WHOI Ocean Life Institute.
Collaborators:
- Zhaohui Aleck Wang, Woods Hole Oceanographic Institution

A New pH Proxy Based on Raman Spectroscopy and its Application to Biocalcification Studies
Biogenic production and sedimentation of calcium carbonate in the ocean, referred to as the carbonate pump, has profound implications for the ocean carbon cycle, and relate both to global climate, ocean acidification and the geological past. Many carbonate-secreting organisms, such as foraminifera and corals, exert strong controls on their biocalcification processes and promote calcification by increasing the pH of their calcifying fluid. We aim to develop a new in vivo pH proxy based on Raman spectroscopy to study the pH regulation inside carbonate-secreting organisms. Our goal is to achieve better understanding of the biocalcification processes in carbonate-secreting organisms to allow improved estimation of marine biogenic carbonate production and predictions of the future environmental-change impacts. Supported by WHOI Interdisciplinary Research program.
Collaborators:
- Frieder Klein, Woods Hole Oceanographic Institution
- Joan Bernhard, Woods Hole Oceanographic Institution
