David Ralston, Dennis McGillicuddy, Don Anderson, Michael Brosnahan, and (WHOI) and Katherine Hubbard (Florida Fish and Wildlife Conservation Commission)
Our goal is to understand and predict how environmental variability influences harmful algal bloom (HAB) dynamics and exposure of human populations to HAB toxins, with implications for human health. We will incorporate existing knowledge along with new insights on populations of the toxic dinoflagellate Alexandrium catenella and the diatom Pseudo-nitzschia spp. into models. The central hypothesis is that environmental changes will influence the composition, distribution, and severity of HABs in ways that are predictable based on response to previous forcing. We will investigate these processes in three different model systems: the Gulf of Maine (GOM), Nauset Marsh (NM), and the coastal Arctic.
Specific Aims:
Aim 1. Gather data to characterize the variability of HABs in the GOM region under past and current environmental forcing conditions.
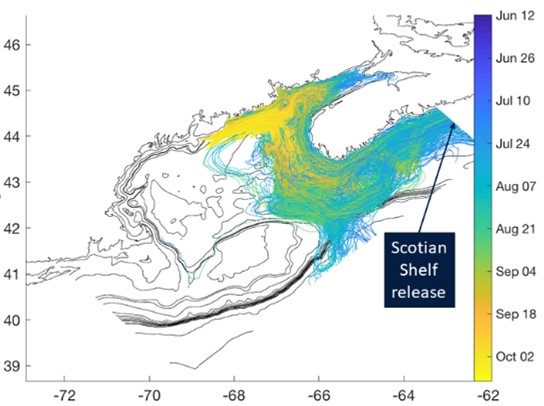
Alexandrium catenella and paralytic shellfish poisoning (PSP) have been public health concerns in the GOM for many decades, while the presence of toxic Pseudo-nitzschia spp. has been noted over a shorter period. We hypothesize that long-term records of toxicity (toxin accumulation in shellfish) due to A. catenella and Pseudo-nitzschia spp. abundance correlate with variability in environmental forcing over this period. We further hypothesize that variability in cell abundance and toxicity in recent and on-going measurements is associated with shifts in water properties, including temperature, salinity, and nutrient concentrations, at both interannual and decadal time scales.
Aim 2. Improve the predictive model for A. catenella and develop a predictive model for Pseudo-nitzschia spp.
WHCOHH research has transformed our understanding of A. catenella in the environment, finding that in situ rates and behaviors differ significantly from laboratory data. We hypothesize that incorporation of this information will markedly enhance model skill for A. catenella in the GOM and help resolve previous inconsistencies with the observational record. Predictive models for Pseudo-nitzschia spp. in the northeast U.S. are not yet available. We will fill this gap by implementing a state-of-the-art biogeochemical and ecosystem model that resolves several functional types of phytoplankton, including diatoms. We will then use knowledge of particularly toxic Pseudo-nitzschia species in the GOM to refine this model to explicitly represent them and their toxin production within the diatom component of the model.
Aim 3. Use toxicity and exposure submodels to assess threats to human health.
The HAB models described above predict the concentrations of harmful algae in the water. Assessment of their impacts on human health requires knowledge of the accumulation of those toxins in shellfish and the degree to which human populations are exposed to the contaminated resource through consumption. Both aspects will be estimated through toxicity and exposure submodels that will be coupled to the HAB population models.
Integration
The proposed research will be integrated into the WHCOHH in several ways: (1) what is learned about bloom dynamics in Project 1 will be used to improve HAB models; (2) development of the exposure submodel will be undertaken in collaboration with Project 3; (3) all three projects will participate in the analysis of environmentally-forced variations in HABs and how they affect human exposure to toxins; and (4) results will be communicated through the Community Engagement Core.
References Cited
Clark, Suzanna, Katherine A. Hubbard, Dennis J. McGillicuddy Jr, David K. Ralston, and Sugandha Shankar. 2021. Investigating Pseudo-nitzschia australis introduction to the Gulf of Maine with observations and models. Continental Shelf Research 228. Elsevier: 104493.